Energy Storage
Our group is focused on investigating the fundamentals of electrochemistry in novel architected electrode materials and electrolytes. Our 3D architected electrodes are designed with full control over the surface area, active material loading, geometry, and size such that it is possible to elicit the desired energy or power density, as well as cyclability and capacitance. With these efforts, we hope to overcome some of the key limitations of energy storage materials such as mechanical failure, sluggish kinetics, and low active material loading.
Current Projects:- 3D Architected LiCoO2 through Gel Infusion and Calcination Additive Manufacturing
- Nanomechanical Behavior of Electrochemically Grown LiCoO2 and Li-based Composite Electrodes
- 3D Architected Lithium Metal Electrodes with Carbon Scaffold
- 3D Architected LiFePO4/C Composite Electrodes
- Catalytic Graphitization of 3D Architected Carbon Electrodes
- 3D Interdigitated Solid-State Lithium-Ion Batteries
- Architected Carbon Flow-Through Electrodes for Selective CO2 Reduction
- Photopolymerizable Polymer Electrolytes for Lithium-Ion Batteries
- Developing Lithium Sulfur Batteries
- Electrochemical Characterization of Architected Electrodes
Current Projects
3D Architected LiCoO2 through Gel Infusion and Calcination Additive Manufacturing
Researcher: Yuchun Sun (Ph.D. student in Materials Science)
LiCoO2 is commonly used as the cathode material of lithium-ion batteries. It stores and releases lithium ions through a reversible intercalation mechanism, which gives great cycling stability to rechargeable batteries. Energy density improvement of conventional LiCoO2 slurry electrodes has been challenging due to the limited electrode thickness, which is in general less than 50 µm to optimize Li ion transport into electrodes. This constraint limits the mass ratio between active material and metal current collector, and low active material loading as a result limits the energy density of the whole battery system. To overcome the dilemma between active material loading and Li ion transport in electrodes, we developed a method for the fabrication of 3D architected LiCoO2 electrodes through gel infusion and calcination. Going from 2D slurry electrodes into 3D architected electrodes, we achieve high active material loading while maintaining Li ion diffusion distance into the electrode material. This method separates the polymer gel 3D printing from cathode material functionalization, which enables the bypass of complicated photoresin design for micro-scale additive manufacturing of a large variety of lithium-ion battery cathode materials.
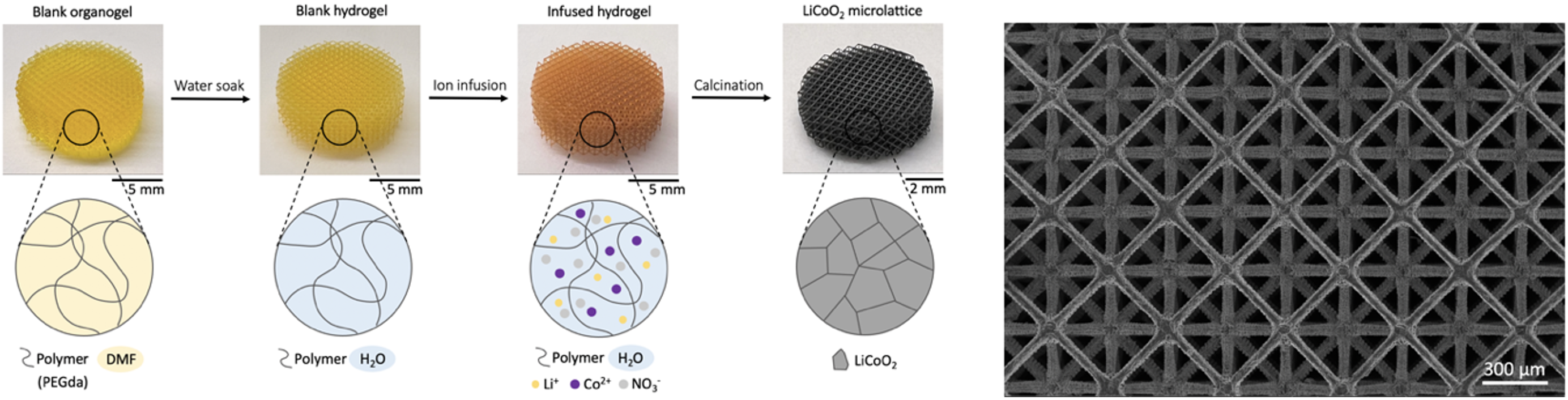
Nanomechanical Behavior of Electrochemically Grown LiCoO2 and Li-based Composite Electrodes
Researcher: Yingjin Wang (Ph.D. student in Materials Science)
Mechanical degradation of cathode active material, such as crack formation and separation of grain boundaries, can lead to isolated active material and longer Li pathways. To prevent capacity fade caused by mechanical failure and provide insight into cathode design, it is important to understand the mechanical response of cathode material and the influence of electrochemical lithiation/dlithiation on material's mechanical property. We utilize in-situ nanoindentation experiment to investigate micro-scale mechanical behavior of electrodeposited LiCoO2 as function of state-of-charge/depth-of-discharge and morphology. The Vacushut and mechanical arm combined with SEM system are used to avoid exposing charged cathode to air. Apart from the cathode, the deformation behavior of air-sensitive Li-based composite anodes is also detected through uniaxial compression of micro-pillars and nanoindentation experiments. Our results help to understand the interfacial morphology between the solid-state electrolyte and metal anode which is highly influenced by the deformation of anode metal.

3D Architected Lithium Metal Electrodes with Carbon Scaffold
Researcher: Yuchun Sun (Ph.D. student in Materials Science), in collaboration with JPL Electrochemical Research, Technology, & Engineering Group
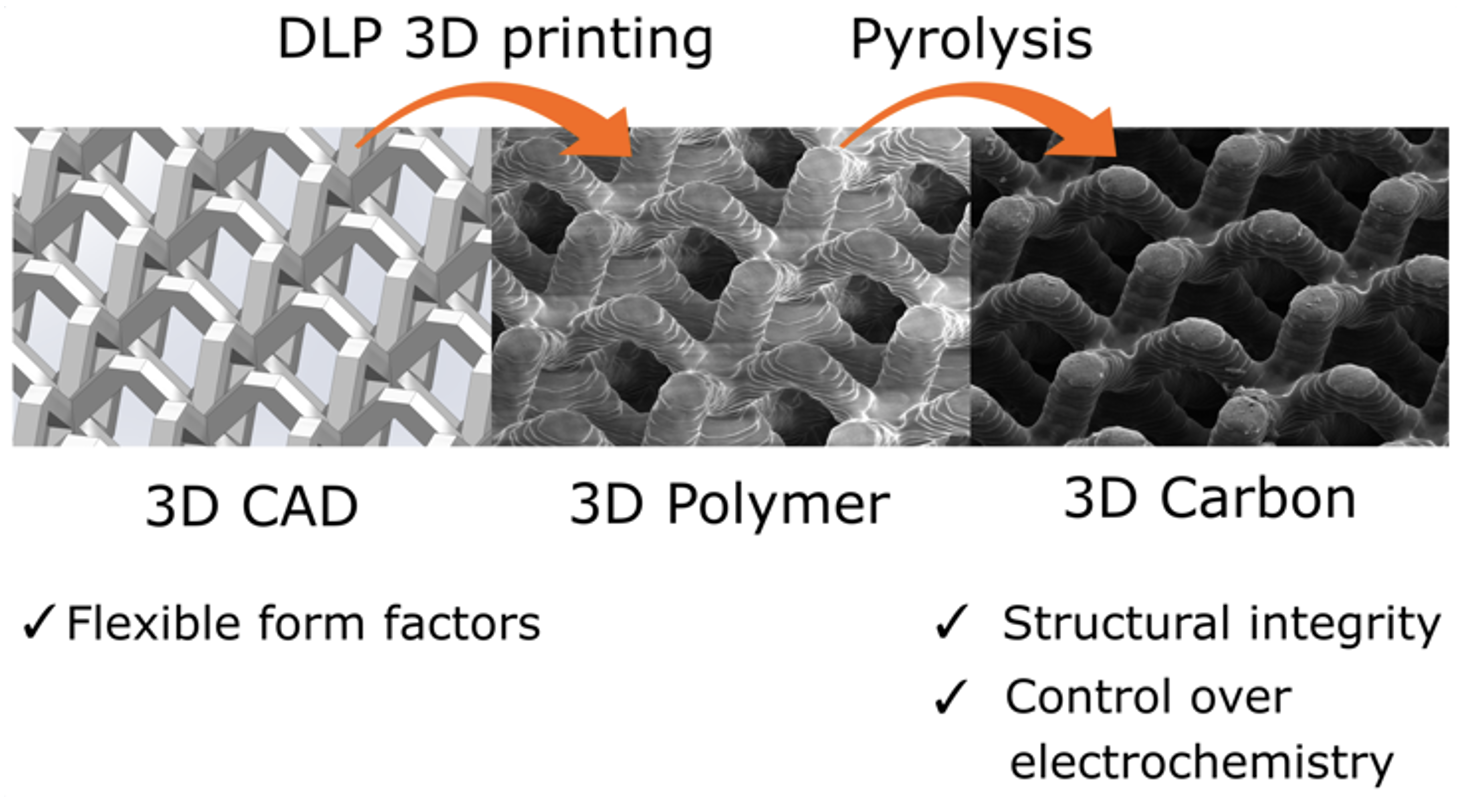
Safe and durable lithium metal electrodes will bring revolutionary increase in battery energy density. Morphology of deposited lithium during lithiation is closely related to current density and ionic distribution near the electrode surface. To lower the effective current density in order to mitigate Li dendrite growth, host materials have been widely studied, among which porous carbon, usually made of carbon fiber, carbon nanotube, graphene or biomass-derived carbon, are most promising due to their outstanding chemical resilience and light weight. We utilize photopolymerization 3D printing and pyrolysis to fabricate 3D architected porous carbon scaffold with flexible micron-to-centimeter form-factors and out-standing mechanical strength. We investigate its surface functionalization and study lithium electrodeposition conditions to achieve dense lithium nucleation and uniform epitaxial growth on the surface of carbon scaffold. The fabricated free-standing 3D architected lithium metal electrode is able to accommodate the large mechanical stress built up during high-rate cycling and keep rounded lithium morphology during high-current operation.
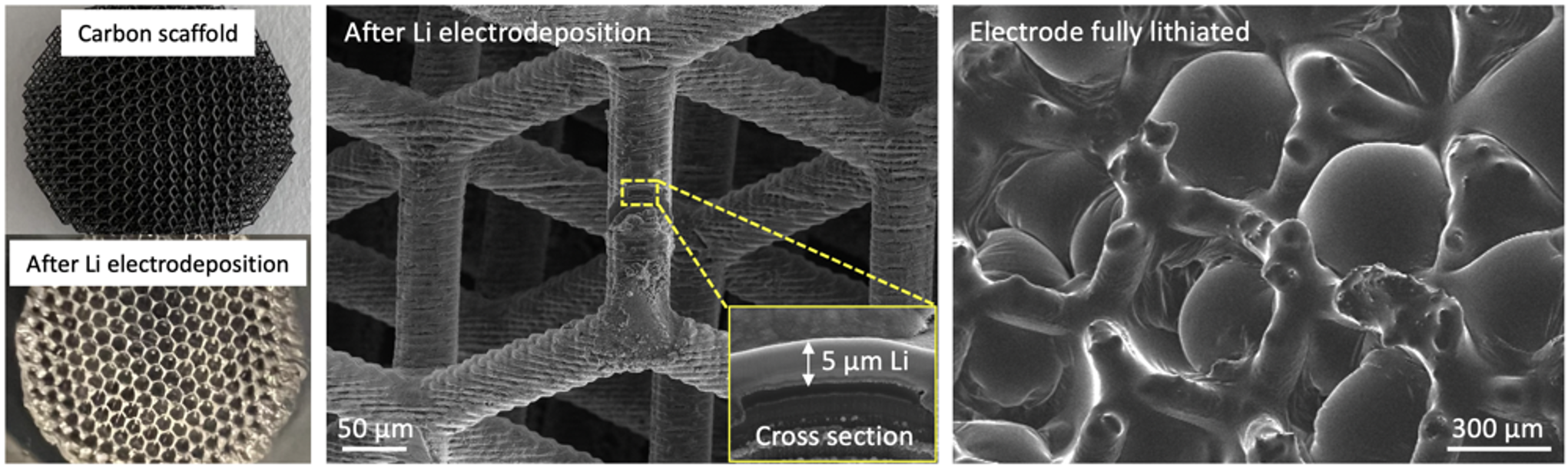
3D Architected LiFePO4/C Composite Electrodes
Researcher: Yingjin Wang (Ph.D. student in Materials Science)
LiFePO4 (LFP) is a commonly used cathode material with high thermal and chemical stability, long cycle life, low cost, and non-toxicity. Two main challenges faced by LiFePO4 as a cathode material are its relatively low ion diffusivity and electrical conductivity. To provide larger interfacial area and improve the active material loading without increasing ion diffusion distance, the LiFePO4/C micro-lattice is fabricated via vat photopolymerization additive manufacturing with high printing resolution. The carbon network in the microlattice which forms in-situ during LiFePO4 synthesis can provide efficient pathways for electron transport to reduce the internal resistance of electrode.

Catalytic Graphitization of 3D Architected Carbon Electrodes
Researcher: Ethan Klein (Ph.D. student in Materials Science)
The inherent geometry of slurry-based graphite electrodes limits researchers' ability to tune key parameters, such as the diffusion length within the electrode, the path of ions in the electrolyte/electrode region, the extent of active material loading, and the overall mechanical strength. By contrast, electrodes with scaffolded 3D structures offer freedom over such tunable parameters. While the tunability of scaffolded electrodes may appear attractive, there exists a tradeoff: current additive manufacturing techniques offer limited control over the crystal phase and microstructure at temperatures below 3000°C. We develop a method for microstructural and crystal phase tuning of scaffolded carbon electrodes via catalytic graphitization. Electrodes are 3D printed using a hygroscopic polymer resin capable of swelling in an aqueous nickel solution, and then pyrolyzed in a vacuum furnace between 800-1000°C, where nickel catalytically produces graphite. XRD patterns display pure glassy carbon at zero nickel concentration, while both glassy and graphitic carbon signatures appear as nickel incorporation increases. Future studies will compare the electrochemical performance of slurry and scaffolded electrodes with varying degrees of graphitic microstructure and architectural features.
3D Interdigitated Solid-State Lithium-Ion Batteries
Researcher: Yuchun Sun (Ph.D. student in Materials Science)
3D lattice electrodes enable high active material mass loadings and high gravimetric capacity of lithium-ion batteries. The idea of 3D batteries goes one step further than 3D lattice electrodes, which not only gives good gravimetric and areal capacities, but also shortens Li ion diffusion distance between two electrodes by making better use of the porous spaces within 3D lattices. Under current fabrication capabilities, 3D battery with interdigitated-plate geometry is one of the most promising approaches. With the gel infusion/calcination-based 3D LiCoO2 fabrication method and the DLP printing/pyrolysis-based 3D carbon fabrication method, we designed and fabricated LiCoO2 and carbon electrodes with interdigitated-plate geometry, which can be inserted into each other with gel polymer electrolytes sandwiched in between. As a natural choice for 3D batteries with complex electrode surface geometries, photopolymerizable gel polymer electrolyte can be easily coated on electrode surface and cured in-situ, giving low electrode-electrolyte interfacial impedance than other solid electrolytes, while eliminating the safety concerns associated with liquid electrolytes.

Architected Carbon Flow-Through Electrodes for Selective CO2 Reduction
Researcher: Yuchun Sun (Ph.D. student in Materials Science), in collaboration with Caltech LiSA Chemical Microenvironments Team
Additive manufacturing enables the fabrication of carbon electrodes with flexible form-factors and well-defined unique geometries. Copper can be deposited on the architected carbon electrodes, allowing for the electrodes to selectively reduce CO2. The hydrodynamics and dissolved species transport of these flow-through electrodes can be modeled and used for the rational design of electrode structure. Using rational electrode design, we want to tune the selectivity of CO2 reduction on copper towards multi-carbon products such as ethylene and ethanol.

Past Projects
Photopolymerizable Polymer Electrolytes for Lithium-Ion Batteries
Researchers: Dr. Fernando Villafuerte (Former Ph.D. student in Materials Science), Yuchun Sun (Ph.D. student in Materials Science)
Replacing graphite with lithium metal as anode material can significantly increase the specific energy density of lithium-ion batteries. Standard electrolytes in use for lithium-ion batteries are composed of organic solvents that are chemically unstable against lithium metal during battery operation. Polymers electrolytes are promising alternatives to avoid catastrophic failure due to combustion of liquid electrolyte, and photopolymerizable electrolyte that cures on electrode surface can lower the interface impedance. We investigate UV curable gel polymer electrolytes with polyethylene glycol system and a novel polyborane-based polymer with Lewis acidic moieties on the backbone. We characterize their conductivity and lithium ion transference number through the use of potentiostatic techniques such as impedance spectroscopy, to investigate how these properties depend on factors such as salt concentration, molecular weight, and amount of cosolvent, aiming to understand the inherent structure-property relationships in order to optimize electrolyte design for potential applications in lithium-ion batteries.

Developing Lithium Sulfur Batteries
Researcher: Dr. Max Saccone (Former Ph.D. student in Chemical Emgineering)
Li-S batteries are poised to outcompete Li-ion batteries in key sectors such as transportation and grid storage due to their use of low cost and earth abundant materials, but improving energy density and mitigating degradation in Li-S batteries is necessary before these applications can be realized. Mechanical degradation mechanisms such as the detachment of insulating Li2S from the conductive matrix which causes irreversible capacity fade are still poorly understood. We investigate the material properties and deformation mechanisms of Li2S via in situ SEM mechanical experiments. We also explore the use of stereolithographic additive manufacturing as a route towards producing mechanically strong and tough 3D architected electrodes with complex architectures, high active material loadings, and large areal capacities relative to conventional 2D film electrodes. We fabricate architected 3D lithium sulfide-carbon composite cathodes with feature sizes as small as 50 µm and a tunable hierarchal internal pore structure, providing a 3-fold improvement in resolution over previously reported extrusion-based additive manufacturing processes for Li-S cathode materials.

Electrochemical Characterization of Architected Electrodes
Researcher: Dr. Kai Narita (Former Ph.D. student in Materials Science)
Understanding the aging mechanisms in battery systems facilitates the development of their effective utilizations, but it is limited by multi-scale dynamics in the stochastic structure of slurry electrodes. To address this challenge, we prescribe the design and fabrication of 3D architected battery electrodes to effectively characterize sources of aging such as solid electrolyte interface (SEI) formation and lithium dendrite growth via in situ visualization and electrochemical monitoring, in addition to post-characterization techniques. Electrochemical phenomena in battery systems are coupled at various scales: namely, at the levels of interfacial reactions and the transport of carriers such as ions and electrons. These behaviors are governed by the distribution of current and potential, which are directly related to structural factors such as mean transport trajectories, electrode surface area, and the relative positions of electrode. By using 3D architected electrodes with fully controllable features and flexible form-factors, we can systematically explore operating batteries to understand their aging mechanisms.
Publications
1. Narita, Kai; Saccone, Max A.; Sun, Yuchun; Greer, Julia R. (2022) "Additive manufacturing of 3D batteries: a perspective" Journal of Materials Research, 37(9), 1535-1546. ISSN: 0884-2914.
2. Saccone, Max A.; Greer, Julia R. (2021) "Understanding and mitigating mechanical degradation in lithium-sulfur batteries: additive manufacturing of Li2S composites and nanomechanical particle compressions" Journal of Materials Research, 36(18), 3656-3666. ISSN: 0884-2914.
3. Narita, Kai; Citrin, Michael A.; Yang, Heng; Xia, Xiaoxing; Greer, Julia R. (2021) "3D Architected Carbon Electrodes for Energy Storage" Advanced Energy Materials, 11(5), Art. No. 2002637. ISSN: 1614-6832.
4. Citrin, Michael A.; Yang, Heng; Nieh, Simon K.; Berry, Joel; Gao Wenpei; Pan, Xiaoqing; Srolovitz, David J.; Greer, Julia R. (2020) "From ion to atom to dendrite: Formation and nanomechanical behavior of electrodeposited lithium" MRS Bulletin, 45(11), 891-904. ISSN: 0883-7694.
5. Xia, Xiaoxing; Afshar, Arman; Yang, Heng; Portela, Carlos M.; Kochmann, Dennis M.; Di Leo, Claudio V.; Greer, Julia R. (2019) "Electrochemically Reconfigurable Architected Materials" Nature, 573(7773), 205-213. ISSN: 0028-0836.
6. Xu, Chen; Ahmad, Zeeshan; Aryanfar, Asghar; Viswanathan, Venkatasubramanian; Greer, Julia R. (2017) "Enhanced strength and temperature dependence of mechanical properties of Li at small scales and its implications for Li metal anodes" Proceedings of the National Academy of Sciences of the United States of America, 114(1), 57-61. ISSN: 0027-8424.
Videos
Lithium dendrites growing from LiPON through copper coatingResearchers: Dr. Michael Citrin (Former Ph.D. student in Materials Science), Dr. Heng Yang (Former Post-doc)
Researchers: Dr. Xiaoxing Xia (Former Ph.D. student in Materials Science), Dr. Heng Yang (Former Post-doc)